
Every species, from bacteria to humans, is capable of regeneration. Regeneration is mediated by the molecular processes that regulate gene expression to control tissue renewal, restoration and growth.
A collaboration between researchers in the Department of Biomedical Engineering and the College of Medicine at Texas A&M University identifies the crucial role of minerals in regulating gene expression, thus controlling the number of proteins that a cell should make, thereby encouraging tissue regeneration and redefining cellular identity.
This research paves the way for future studies to identify the role of specific minerals, as well as how they can be assembled to design the next generation of mineral medicine to heal damaged tissue.
This study was recently published in Science Advances.
Minerals are inorganic elements that play many vital roles, working interactively with vitamins, enzymes, hormones and other nutrient cofactors to regulate thousands of the body’s biological functions. Although several minerals have been shown to regulate gene expression and cellular activity, very little work has focused on understanding underlying molecular mechanisms.
This engineering research group is led by Dr. Akhilesh Gaharwar, associate professor of biomedical engineering and Presidential Impact Fellow, in collaboration with Dr. Irtisha Singh, assistant professor in the Department of Molecular and Cellular Medicine at Texas A&M and the co-corresponding author of the study where a new class of mineral-based nanoparticles has been introduced to direct human stem cells toward bone cells. These nanoparticles are known specifically as nanosilicates, and with them, the team is able to determine the role of minerals in regulating gene expression profiles to direct stem cell differentiation.
These nanosilicates are disc-shaped mineral-nanoparticles 20-30 nanometers (nm) in diameter and 1-2 nm in thickness. These nanoparticles are highly biocompatible and are readily eaten up by cells. Once inside the cell body, these nanoparticles slowly dissolve into individual minerals such as silicon, magnesium and lithium.
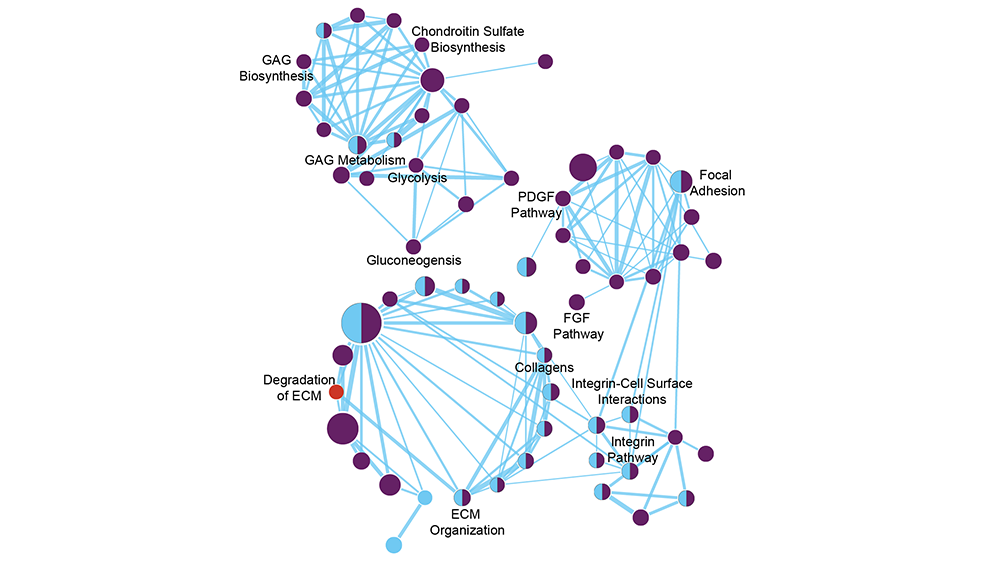
Nanosilicates dissociate into individual minerals inside the cells and turn “on” a set of key genes that result in information flow throughout the cells, known as signaling pathways. These signaling pathways are responsible for instructing the cells to take on specific functions, such as converting into another type of cells or starting the healing process by secreting tissue-specific proteins known as extracellular matrix.
These extracellular matrices are composed of various proteins, including glycoproteins and proteoglycans that facilitate tissue healing and support tissue functions.
Combining interdisciplinary techniques and biomedical engineering and genomics methods, the lead authors of this study, doctoral students Anna Brokesh and Lauren Cross, identify and characterize significant genes that are turned “on” and activated by different signaling pathways due to treatment with minerals. One of the major findings of this study is that minerals such as silicon, magnesium and lithium are involved in inducing endochondral ossification, a process by which stem cells are transformed into soft and hard tissues such as cartilage and bone in young humans.
The Singh Laboratory, managed by Singh, leverages high-throughput functional assays and perturbations to dissect the functional regulatory programs in mammalian cells.
In this study, they analyzed whole transcriptomic sequencing (RNA-seq) data to evaluate the effect of nanosilicates and ionic dissolution products on the gene expression profiles of stem cells. RNA-seq, a transcriptome-wide high throughput sequencing assay, provides an unbiased and holistic overview of the gene expression profiles to identify pathways that are perturbed by specific treatments.
“There are a lot of people who want to understand how minerals impact the human body, but there is limited evidence to identify how they affect us on the cellular level,” Brokesh said. “Our study is one of the first studies to utilize unbiased transcriptome-wide sequencing to determine how mineral ions can direct stem cell fate.”
The proposed approach addresses a long-standing challenge in current therapeutic approaches that utilize supraphysiological doses of growth factors to direct tissue research. Such a high dose of growth factors results in a range of complications, including uncontrolled tissue formation, inflammation and tumorigenesis, the production or formation of tumor cells. These adversely limit the usage of growth factors as a therapeutic agent in the field of regenerative medicine.
Gaharwar said the impact of this work is far-reaching because understanding the effect of minerals to achieve desired regulation of cellular activity has a strong potential to open novel avenues for developing clinically-relevant therapeutics for regenerative medicine, drug delivery and immunomodulation.
This study was funded by the National Institute of Biomedical Imaging and Bioengineering, the National Institute of Neurological Disorders and Stroke, and the Texas A&M University President’s Excellence Fund.
Other authors who contributed to this study are graduate researchers Anna L. Kersey and Aparna Murali, undergraduate researcher Christopher Richter, and Dr. Carl Gregory, associate professor of molecular and cellular medicine in the College of Medicine.